![IMG_4490[1]](https://biobasedorgchem.com/wp-content/uploads/2024/03/IMG_44901-scaled.jpg)
| Jay Hanssens | |
| PhD Student | |
| Room: | 200.3.049 |
| Email: | j.hanssens@maastrichtuniversity.nl |
| LinkedIn: | https://www.linkedin.com/in/jay-hanssens-a47a87114/ |
Isocyanide-based multicomponent peptide chemistry
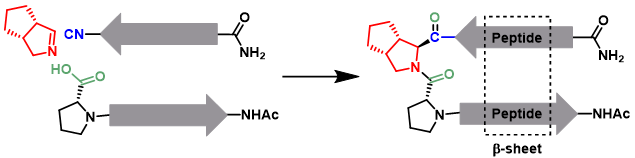
For many years, chemists explored the world of amino acids, peptides and proteins. Aside from the fact that these molecules are fully bio-based and strongly abundant, they can also be utilized in the development of medicinally relevant structures. The development of novel chemical methodologies with these compounds can therefore be of great significance to society. As our group is specialized in isocyanide-based multicomponent chemistry, Jay is seeking to combine this field of research with that of amino-acid/peptide chemistry. Multicomponent reactions like the Passerini or the Ugi provide interesting routes to chemically modify peptides or synthesize peptidomimetics. An example is the β-turn, a secondary structural feature that is now gaining more interest amongst peptide chemists for its applicability pharmaceutical peptide synthesis. A template that has extensively been used for this purpose is D-Pro-L-Pro to provide antiparallel β-sheets. Conventionally, chemists installed this secondary structural feature by means of SPPS. However, this method is low yielding, highly wasteful and can merely create peptides of up to approximately 50 amino acids. Here we propose a novel method for the synthesis of such D,L-proline based β-turns to facilitate the formation of β-sheets. The Ugi multi-component reaction is an ideal method for the synthesis of a D-proline-L-proline site in a single step. By employment of a chiral imine well-known to our group, a selective Ugi reaction should successfully result in the involved β-turn.2 In conventional methods such as solid phase peptide synthesis (SPPS), the coupling of a proline onto another proline is usually difficult and results in a lower yield. By utilizing our multicomponent approach the difficulty of attain this structural feature is greatly diminished and could result in much higher yields.